A new line of research suggests that human fertility depends on complex immunological mechanisms that, until recently, were scarcely explored in conventional medicine.
If this dimension is confirmed, infertility could cease to be considered solely a hormonal or anatomical issue and instead be understood as a matter of immune regulation.
By Ehab Soltan
HoyLunes — For decades, infertility has been analyzed primarily through three prisms: hormonal, anatomical, and genetic. The search focused on obstructed fallopian tubes, ovulation imbalances, or chromosomal abnormalities. However, despite possessing advanced diagnostic technology, doctors worldwide often face an enigma: “idiopathic sterility,” or the inability to conceive without an apparent cause.
The magnitude of the problem justifies the growing scientific interest. According to the World Health Organization (WHO), one in six people worldwide experiences infertility at some point. What is unsettling is that in a significant percentage—between 10% and 30% of cases—specialists find no clear medical explanation. This diagnostic void has prompted a provocative question: What if a substantial portion of these cases were, in reality, an undetected immunological disorder?
Our immune system is much more than a defense force against pathogens. It acts as an ultra-sensitive regulator of biological processes, where pregnancy represents nature’s most ambitious “diplomatic project”. A harmonious interaction between the maternal organism and the embryo is not a matter of chance, but the result of subtle molecular negotiations.

Pregnancy: An Extraordinary Immunological Challenge
From the perspective of classical immunology, pregnancy is a paradox. The embryo carries the father’s genetic signature, making it a “semi-allograft”: a tissue that is 50% foreign to the maternal body.
Normally, the immune system would eliminate any cell not identified as “self.” However, during gestation, the miracle of maternal-fetal immunological tolerance occurs. The mother must not only tolerate the embryo but actively support it without lowering her guard against actual infections. When this “diplomacy” fails, the body begins to treat the guest as an intruder.
During implantation, the immune system undergoes a remarkable reorganization. Some cells reduce their cytotoxic (attack) activity, while others adopt regulatory functions. This process involves molecular interactions between the endometrium and the embryo that are currently the focus of research at the most advanced reproductive hubs in the world.
When the Immune System Becomes an Obstacle
Research demonstrates that immunological dissonances can sabotage the reproductive process at critical stages:
Failed Landing (Implantation):** Imbalances that block the initial “dialogue” necessary for the embryo to nest.
Silent Fire (Endometrial Inflammation):** Chronic and imperceptible inflammatory processes that make the uterus uninhabitable.
Early Rejection: Aggressive reactions that cause gestational loss before the pregnancy is clinically detectable.
Autoimmune Factors: Pre-existing disorders where the body is programmed to attack its own tissues, increasing the risk of miscarriage.
Recent studies explore the relationship between inflammatory markers and recurrent implantation failure. Although not conclusive, they suggest that certain immunological profiles drastically reduce the chances of success, even in assisted reproduction techniques.
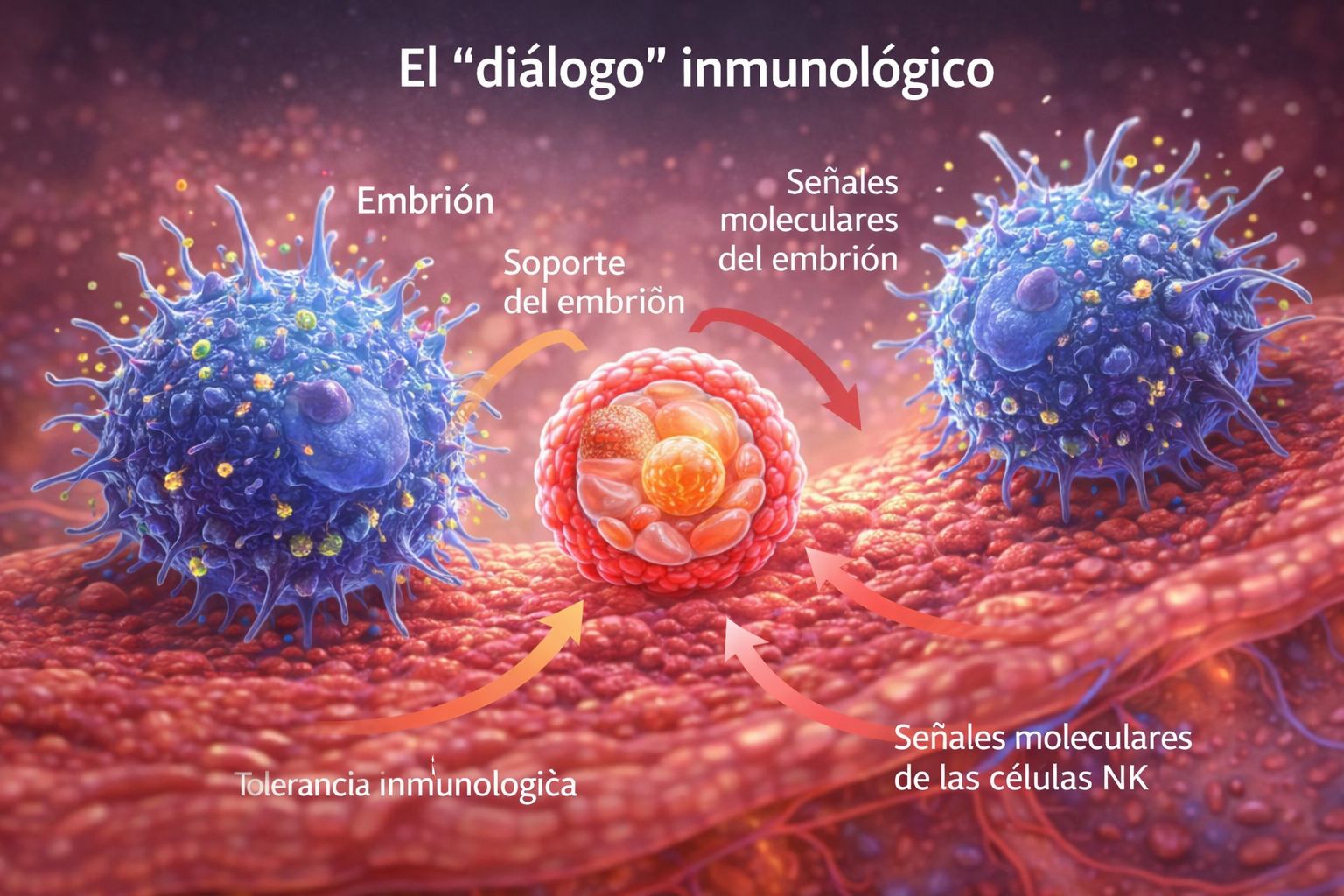
The Guardians of the Uterus: Uterine NK Cells
One of the most relevant discoveries is the role of uterine Natural Killer (uNK) cells. Unlike their relatives in the bloodstream, whose function is to eliminate infected or tumor cells, uNK cells in the uterus have a constructive mission:
They regulate the depth of implantation.
They control the remodeling of maternal blood vessels to nourish the embryo.
They support the early development of the placenta.
In the early endometrium, these cells can constitute up to 70% of the local immune cells, confirming that they are key architects of the uterine environment. An imbalance in their activity—whether through excess aggression or a lack of support—can seal the future of the pregnancy.
The Future of Reproductive Medicine: Between Hope and Caution
If this hypothesis is consolidated, we are facing a paradigm shift that would include:
Precision Diagnosis: Immunological profiles integrated into routine check-ups.
Targeted Immunomodulation: Therapies that foster uterine tolerance without suppressing general defenses.
Holistic Approaches: Treating systemic inflammation as part of the fertility protocol.
Nonetheless, the scientific community calls for prudence. The field is complex and, at times, controversial. Many proposed treatments still lack solid clinical evidence, and societies such as ESHRE or ASRM warn of the need for more large-scale trials before standardizing these therapies in routine practice.

Toward a New Reproductive Sovereignty
For decades, fertility was a matter of mechanics and chemistry. Immunology now adds a deeper, almost philosophical dimension: fertility is not a static state, but a dynamic equilibrium.
The challenge of the 21st century is not only to stimulate hormones or perfect laboratory techniques but to understand the triad of immunity, inflammation, and reproduction. The question that remains open is as simple as it is profound: How many cases of infertility remain invisible because we do not yet know how to listen to the body’s immunological language?
Scientific Support and Sources:
National Institutes of Health (NIH): [www.nih.gov]
Nature / The Lancet: Studies on uNK cells and inflammatory markers.
ESHRE / ASRM: Consensus documents on idiopathic sterility.
#ImmunologicalInfertility #ReproductiveMedicine #WomensHealth #MedicalInnovation #HoyLunes #EhabSoltan



